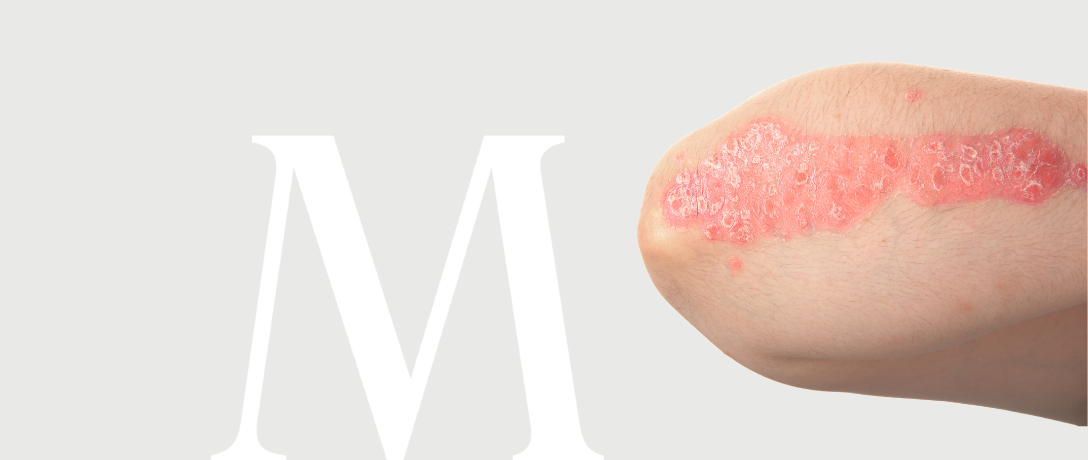
Dermatology – pustular psoriasis
DermatologyRandomized, double-blind, placebo-controlled phase 3 study to evaluate the efficacy and safety of an investigational medicinal device in the treatment of adult patients with generalized pustular psoriasis.
More information:
Patients 18-80 years of age with a confirmed clinical diagnosis of generalized pustular psoriasis or experiencing a first episode of the disease without concomitant form of psoriasis can participate in the study.
For your participation in the study, you receive:
- qualifying visit,
- control visits to assess the effectiveness of treatment,
- a wide panel of diagnostic and control tests,
- medical care related to the study,
- 4 weeks of treatment, with a possible extension to 8 months,
- an outstanding dermatologist as an attending physician.
Start of study recruitment: December 2021